Imagine taking a medication for years that keeps your condition stable, only to have your doctor switch you to a generic version to save money. Suddenly, your symptoms return, or worse, you experience a severe reaction. You might assume your illness is progressing, but the real culprit could be the pill itself. This is therapeutic failure, a situation where a drug doesn't produce the expected clinical response. While most generics are safe, some fail in ways that can be life-threatening, especially when the margin between a helpful dose and a toxic one is razor-thin.
| Factor | What Happens | Risk Level |
|---|---|---|
| Bioequivalence Gap | Absorption differs by up to 20% from brand name | Moderate to High |
| Manufacturing Errors | Inconsistent active ingredient distribution | High |
| API Degradation | Chemical breakdown due to light or humidity | Moderate |
| Inactive Ingredients | Fillers change how the drug dissolves | Moderate |
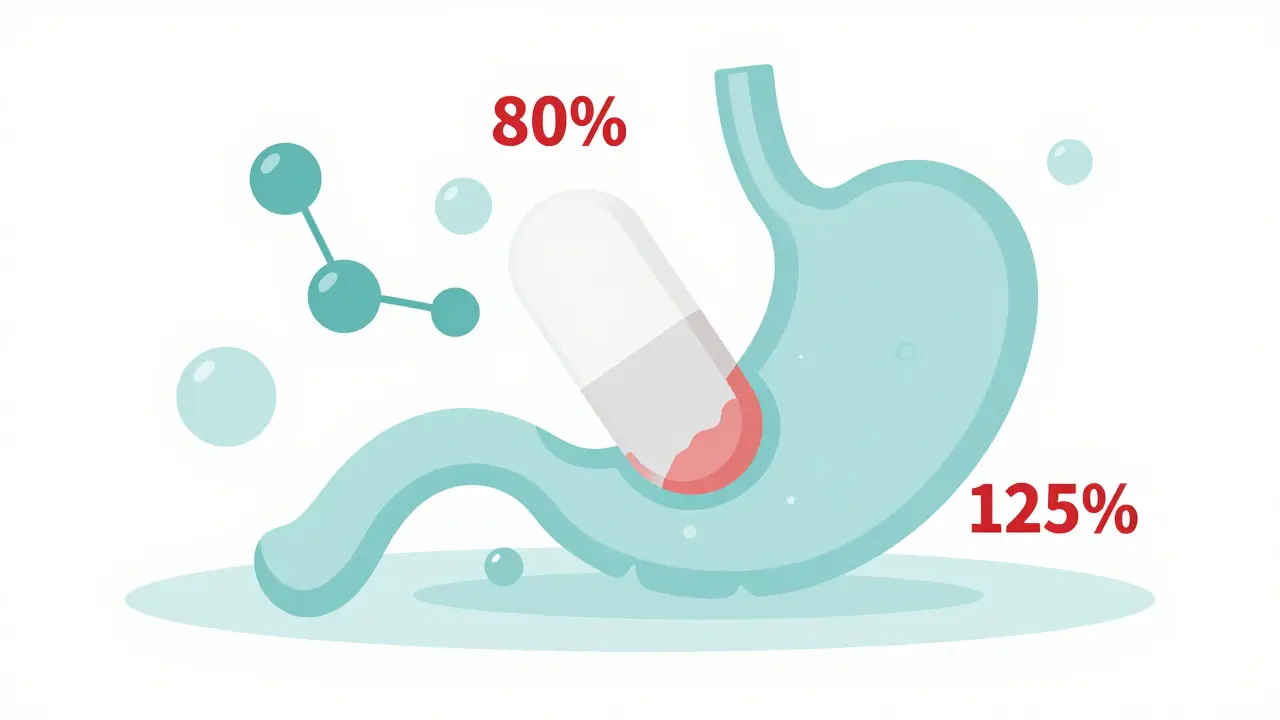
The Bioequivalence Gap: The 80-125% Rule
To get a generic drug approved, the FDA is the federal agency responsible for regulating food and drugs in the United States. They require generics to show bioequivalence, meaning the generic drug must deliver the same amount of active ingredient to the bloodstream at a similar rate as the brand-name version. However, there is a catch: the acceptable range for the area under the curve (AUC) and maximum concentration (Cmax) is typically between 80% and 125%.
Does that sound a bit broad? It is. Theoretically, a generic could provide 20% less medication than the original. For a common headache pill, this might not matter. But for medications with a Narrow Therapeutic Index (or NTI), which is a range of drug concentrations in the blood where the difference between a therapeutic dose and a toxic dose is very small, that 20% gap is a disaster. If you are taking Warfarin to prevent blood clots, a slight dip in efficacy could lead to a stroke, while a slight increase could cause internal bleeding.
When the "Recipe" Goes Wrong
A drug isn't just the active ingredient; it's a combination of that ingredient and various inactive fillers and binders. These "inactive" ingredients aren't always neutral. They can change how a pill dissolves in your stomach. For example, the FDA had to withdraw approval for Budeprion XL (a generic version of Wellbutrin) because the inactive ingredients altered the drug's performance so much that it didn't work like the original.
Manufacturing inconsistencies are a massive part of the problem. Data shows that about 31% of deficiencies in Abbreviated New Drug Applications (ANDAs) are tied to manufacturing issues. This includes poor stability testing or inconsistent distribution of the drug within the tablet. In some horrifying cases documented by the Therapeutic Investigations Bureau of Journalism, chemotherapy drugs used for breast and leukemia cancers contained so little active ingredient that taking them was practically useless, giving the cancer a window to grow unchecked.
The Silent Threat of API Degradation
The Active Pharmaceutical Ingredient (or API) is the chemically active part of a drug that produces the intended pharmacological effect. This chemical can break down long before the expiration date. Hydrolysis occurs when moisture gets into ester-containing APIs, while oxidation and photolysis happen when drugs are exposed to oxygen or light.
When the API degrades, the potency drops. Some quality tests have found drugs containing less than 88% of the labeled amount. Conversely, some contain over 112%. In a study by Dr. Okuda, multiple sclerosis patients who remained stable were using generics with doses between 97.4% and 103.3%. Meanwhile, those who suffered relapses were taking generics that contained as little as 72.5% of the required dose. This isn't just a statistical variance; it's a direct hit to patient health.
Real-World Consequences and Safety Monitoring
Therapeutic failure doesn't always look like a drug "not working." Sometimes it looks like a sudden, toxic overdose. In May 2024, Glenmark Pharmaceuticals recalled 47 million doses of potassium chloride because the tablets didn't dissolve properly. If a drug doesn't dissolve at the right speed, it can "dump" too much medication into the system at once. A heart transplant patient reported concerns that her medication was hitting her system too quickly, which could potentially lead to organ rejection, fatigue, or heart failure.
Safety monitoring is the only way to catch these issues. We've seen high-profile cases like the Valsartan recalls, where nitrosamine contaminants-potential carcinogens-were found in blood pressure medications. The scary part is that we only catch the ones that get tested. There's a systemic lack of transparency in the global supply chain, especially in countries with loose regulatory oversight, where the same blister pack might contain pills with wildly different amounts of active ingredients.

How to Spot and Manage Generic Failure
If you suspect a generic isn't working, the first step is medication reconciliation. This means comparing exactly what you were taking when you were stable versus what you are taking now. Don't assume your disease is simply getting worse. If you notice a sudden shift in your condition after a pharmacy switch, bring the actual bottle to your doctor.
For those on NTI drugs, the standard for bioequivalence should be much tighter-ideally 90-111% instead of 80-125%. If you are on a high-risk medication, ask your pharmacist if the generic is an "NTI-grade" formulation or if there are known stability issues with that specific manufacturer. In some cases, the only safe move is to request the brand-name version through a "dispense as written" (DAW) order.
Are all generic drugs dangerous?
No. The vast majority of generics are bioequivalent and work exactly like brand-name drugs. The risk is primarily concentrated in drugs with a Narrow Therapeutic Index (NTI) or those from manufacturers with poor quality control.
How can I tell if my generic drug is failing?
Look for a sudden return of symptoms after a brand switch, new and unexpected side effects, or a lack of response to a dose that previously worked. Always consult your doctor before changing your medication.
What is the 80-125% rule?
It is the FDA's standard for bioequivalence, meaning a generic drug's absorption (AUC and Cmax) can be 20% lower or 25% higher than the brand-name drug and still be approved.
Why do inactive ingredients matter?
Inactive ingredients (fillers, binders) affect the drug's dissolution rate. If a pill dissolves too fast or too slow, it can lead to toxicity or therapeutic failure, even if the active ingredient amount is correct.
What should I do if I suspect a therapeutic failure?
Contact your healthcare provider immediately. Provide them with the exact manufacturer and lot number from your medication packaging so they can track the issue and potentially switch you back to the brand-name version.
Next Steps for Patients and Providers
If you are a patient, keep a log of when you switch generic manufacturers. Note any changes in how you feel within the first two weeks of a new prescription. If you are a provider, be skeptical of "sudden progression" in stable patients who have recently changed pharmacies. Prioritizing transparency and demanding tighter bioequivalence for high-risk drugs is the only way to prevent these failures from becoming fatalities.



