Tag: interchangeable biosimilars
The Purple Book: Understanding Biosimilars and Interchangeability from the FDA
- Benjamin Aghaki-Allen
- Health
- 15 comment
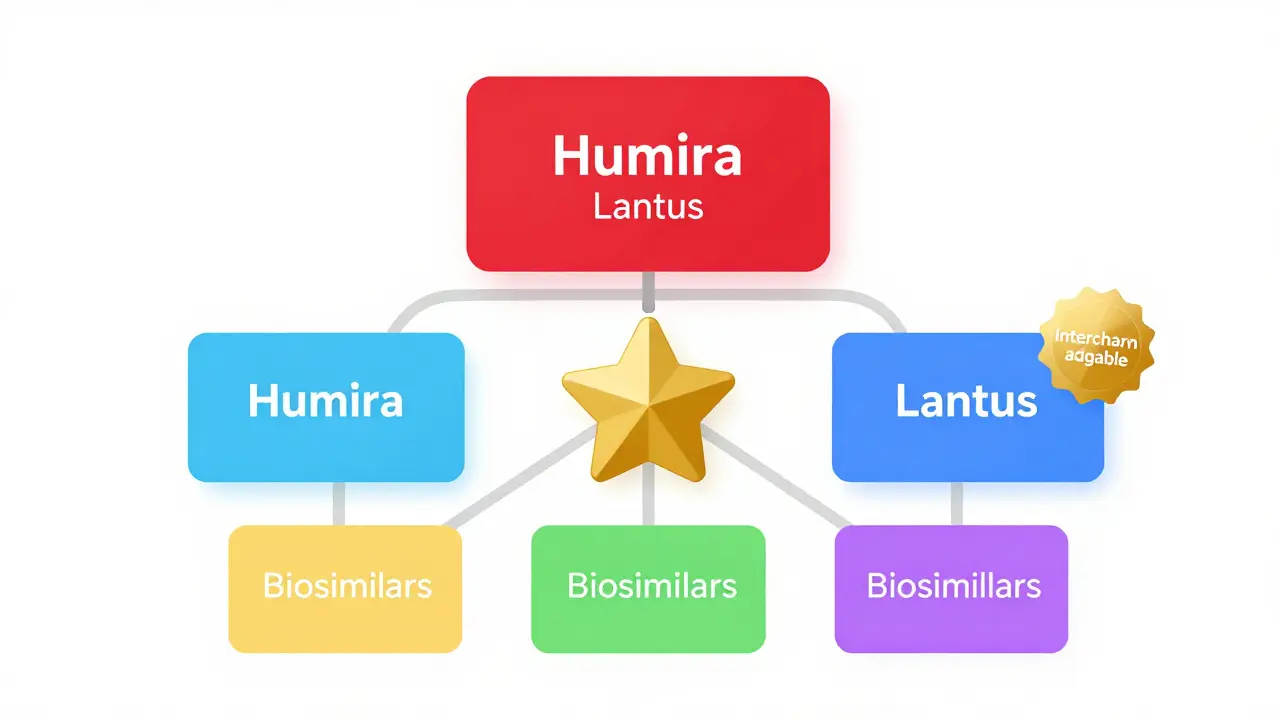
The FDA's Purple Book is the official guide to biosimilars and interchangeable biological products. Learn how it helps patients and pharmacists understand substitution rules, regulatory status, and key differences between biosimilars and interchangeable versions.
VIEW MORECategories
- Health (173)
- Health & Wellness (7)
- Pharmaceutical Research (5)
- Nutrition & Weight Loss (4)
- Climate & Environment (1)
Popular posts
-
Neem: The Super-Plant Transforming Modern Dietary Supplements
Benjamin Aghaki-Allen -
RNA & DNA Supplements: Boost Your Genes for Better Health
Benjamin Aghaki-Allen -
Mast Cell Activation: Understanding Mediator Release and Stabilizer Therapy
Benjamin Aghaki-Allen -
Chronic Hepatitis C: How Modern Antivirals Cure the Virus and Protect the Liver
Benjamin Aghaki-Allen -
Buy Cheap Generic Abilify Online - Safe & Affordable Options
Benjamin Aghaki-Allen
Popular tags
- medication safety
- medication adherence
- side effects
- generic drugs
- online pharmacy
- medication errors
- drug side effects
- bioequivalence
- drug interactions
- dietary supplement
- gut health
- blood pressure medication
- alternatives
- senior medications
- Hatch-Waxman Act
- generic medications
- adverse drug reactions
- medication reconciliation
- generic substitution
- narrow therapeutic index