Imagine your immune system has a set of high-alert sentinels that are a bit too eager. In some people, these cells don't just wait for a dangerous pathogen; they trigger a full-scale alarm over a piece of clothing, a specific food, or even a stressful phone call. This is the core of Mast Cell Activation Syndrome is a condition where mast cells inappropriately release inflammatory mediators, causing multi-system symptoms. Also known as MCAS, it transforms the body's defense system into a source of chronic illness. While the symptoms can feel like a random collection of allergies, the root cause is a biological "misfire" in how these cells release their chemical payloads.
The Sentinels: What Are Mast Cells?
Before we get into the therapy, we need to understand the players. Mast Cells are specialized immune cells of hematopoietic origin that act as sentinels at the body's interfaces, such as the skin, lungs, and gut. First described by Paul Ehrlich in 1878, these cells are essentially chemical warehouses. They are packed with granules-tiny sacs containing potent chemicals-just waiting for the signal to open the floodgates.
In a healthy person, mast cells help fight off parasites and initiate wound healing. But in someone with MCAS, these cells are "twitchy." They react to stimuli that shouldn't be triggers, leading to a cascade of symptoms that can range from hives and flushing to severe anaphylaxis. Interestingly, about 30% of people with this condition have genetic mutations in genes like KIT or TPSAB1, which essentially rewires the cell's "on switch."
The Chemical Payload: Pre-formed vs. Newly Synthesized Mediators
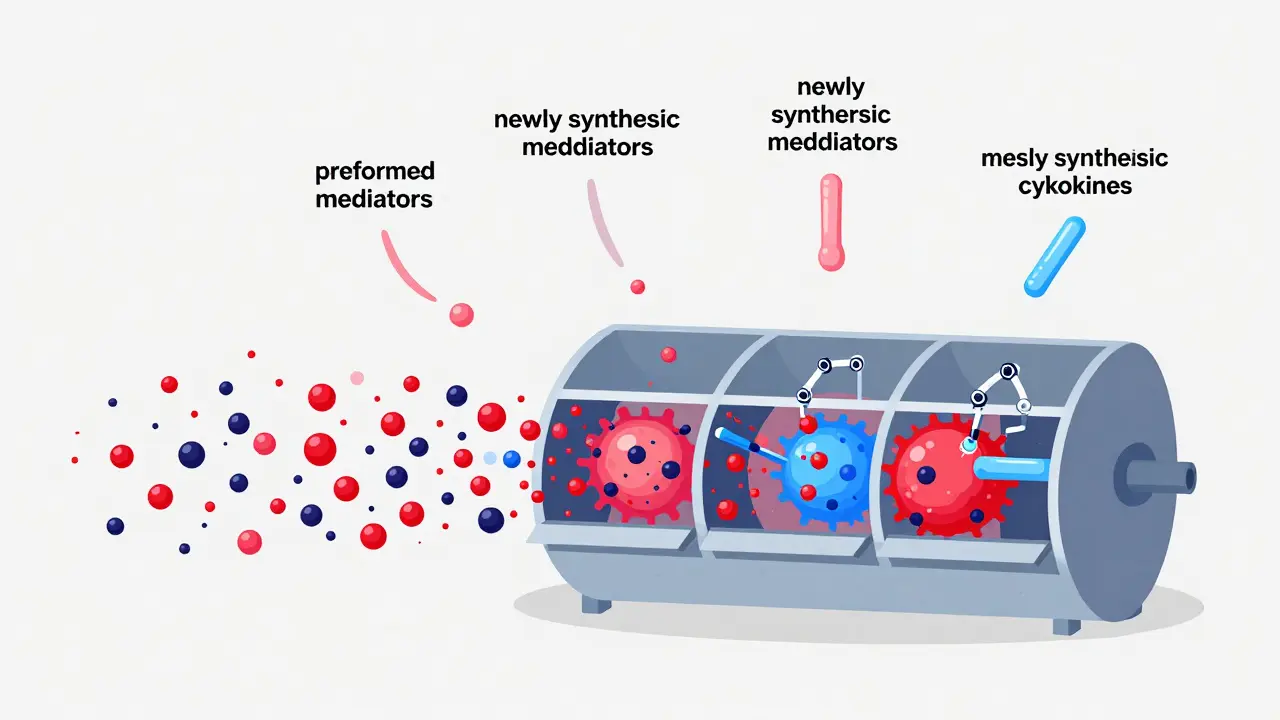
When a mast cell "activates," it doesn't just release one thing; it dumps a complex cocktail of chemicals. Scientists divide these into two main groups based on how they are made and how fast they hit your system.
First, there are the pre-formed mediators. These are stored in granules and released via exocytosis-basically, the cell bursts its bubbles. This happens in seconds. Histamine is the most famous of these, detectable within 15 to 90 seconds of stimulation. Then there's Tryptase, an enzyme that is often used as a biomarker to diagnose MCAS because it's so specific to mast cells.
Then come the newly synthesized mediators. These aren't sitting in a warehouse; the cell has to build them on the fly. Lipid-derived compounds like Prostaglandin D2 (PGD2) and leukotrienes take a few minutes to appear. Finally, cytokines-the signaling proteins that tell other immune cells to join the fight-can take several hours to reach peak levels. This chronological release is why some reactions seem to happen in waves: the "hit" of histamine happens first, followed by the lingering inflammation of cytokines.
| Mediator Type | Examples | Release Time | Primary Function |
|---|---|---|---|
| Pre-formed | Histamine, Tryptase | Seconds to Minutes | Immediate allergic response, itching |
| Lipid-derived | Prostaglandin D2, LTC4 | Minutes | Bronchoconstriction, vascular permeability |
| Cytokines | TNF-α, IL-6 | Hours | Long-term inflammation, immune recruitment |
How Activation Happens: The Triggers
What actually pushes the button? For about 70% of people, it's an IgE-mediated response. This is your classic allergy: an antibody called IgE binds to the cell surface, and when an allergen hits that antibody, the cell dumps its load. But MCAS is trickier. It can be triggered by non-IgE pathways too.
Common non-immune triggers include physical stimuli like heat or pressure, emotional stress, and even certain medications. Many patients use a "trigger wheel" to track their specific sensitivities. For instance, NSAIDs (like ibuprofen) and alcohol are common culprits, affecting over 60% of patients in some surveys. Even microbial products from bacteria can activate these cells through Toll-like Receptor 2 (TLR2), which is why some people feel "allergic" during a cold or sinus infection.
Stabilizer Therapy: Stopping the Leak
Most people are familiar with antihistamines, but those are like trying to mop up a flood while the faucet is still running. They block the effect of histamine, but they don't stop the cell from releasing other chemicals like tryptase or leukotrienes. Mast Cell Stabilizers are medications that prevent the mast cell from degranulating in the first place by stabilizing the cell membrane and blocking calcium influx.
Cromolyn Sodium is the gold standard here. It doesn't treat an active attack; instead, it's a prophylactic. It essentially "locks" the granules inside the cell. If you're taking it correctly, you're preventing the explosion before it happens. However, it's not a magic bullet. It has a short half-life (about 1.5 hours), which is why it's often taken four times a day. It also has a reputation for tasting terrible, which can be a real struggle for pediatric patients.
Another option is Ketotifen. This drug is a bit of a multitasker-it stabilizes mast cells and also blocks H1 receptors. In some studies, it has shown 50-70% efficacy in reducing MCAS symptoms. While these stabilizers are great for baseline control, they often need to be paired with other treatments because they don't stop the production of late-stage cytokines.

Practical Tips for Starting Therapy
If you've just been diagnosed, don't expect overnight results. The journey to a diagnosis is often long-some patients spend 3 to 5 years visiting different doctors before getting an accurate answer. Once you start stabilizers, the titration process is key. Most doctors start with a low dose of cromolyn (100 mg four times daily) and gradually increase it over 4 to 6 weeks to avoid gastrointestinal side effects like nausea or diarrhea.
How do you know if it's working? You can't always rely on how you feel, as symptoms fluctuate. Specialists often monitor 24-hour urinary markers. A reduction of 30% or more in N-methyl-β-hexosaminidase or methylhistamine levels usually indicates the therapy is hitting the mark. Keep a detailed log of your triggers and your response to the medication; this data is gold for your immunologist.
The Future of MCAS Treatment
We are moving beyond simple stabilizers. New biologics like Omalizumab target IgE directly, offering a 70-80% response rate in some cases-significantly higher than stabilizers alone. For more severe cases, such as systemic mastocytosis, targeted therapies like Avapritinib are now being used to target specific genetic mutations (like KIT D816V).
The next frontier involves SYK kinase inhibitors. These are currently in Phase II trials and aim to shut down the internal signaling pathway that tells the cell to release mediators. If these succeed, we could see symptom control rates jumping to 80-90% by 2030, moving us from "managing" the condition to truly controlling it.
What is the difference between an antihistamine and a mast cell stabilizer?
Antihistamines block the histamine receptors on your cells so you don't feel the symptoms (like itching or sneezing). Mast cell stabilizers, however, prevent the mast cell from releasing histamine and other chemicals in the first place. Think of antihistamines as a shield and stabilizers as a lock on the door.
Can I take cromolyn sodium during an acute allergic reaction?
No. Mast cell stabilizers are prophylactic, meaning they must be in your system before the trigger occurs. They are not effective for treating an active, acute reaction. For those situations, fast-acting treatments like epinephrine or antihistamines are required.
Why does it take so long to see results with stabilizer therapy?
Stabilizers work by altering the stability of the cell membrane over time. Many patients report it taking up to 8 weeks of consistent dosing to reach a full therapeutic effect, as the body adjusts and the baseline level of mast cell "twitchiness" decreases.
What are the most common side effects of cromolyn sodium?
The most common issues are gastrointestinal, including nausea and diarrhea, affecting about 35% of users. This is why doctors recommend a gradual titration of the dose over several weeks.
How is MCAS diagnosed if symptoms are so vague?
Diagnosis usually involves a combination of clinical history (multi-system symptoms) and biomarker testing. This includes checking for a significant rise in serum tryptase (often ≥20% above baseline) and testing urinary metabolites like N-methyl-β-hexosaminidase.

This is so helpful! 🌟 It's good to see a clear explanation of how the meds work. 💊
funny how everyone just trusts these pharma pills without questioning the laeger meaning of sickness
probably just a way to make us depndent on a system that doesnt care about the soul
stop overcomplicating this stuff just follow the titration schedule and stop complaining about the taste of the medicine you need it to survive
Oh wow, a drug that takes eight weeks to work and tastes like garbage. Truly a medical miracle we're living in! 🙄
Wake up people! Why are we talking about "stabilizers" when these are just tools to keep us sedated and compliant while they track our immune responses for some larger experiment!! The a-2 language codes and these specific genetic mutations are just a cover for something way more sinister happening in the labs!
cromolyn is basic but effective if you stick to it
Honestly most people fail at this because they lack the discipline to track their triggers properly. I've seen it a thousand times. You can't just take a pill and expect the world to change if you're still eating junk and ignoring your environment. It's simple biology. If you don't have a trigger wheel, you're basically guessing. Most of you just want a quick fix without doing the actual work of healing your own body through strict adherence to the protocol. I know exactly how this works because I've mastered the process of systemic inflammation management. Stop looking for shortcuts and start acting like a patient who actually wants to get better instead of someone who just wants to complain about side effects. It's honestly pathetic how many people give up on stabilizers just because of a little nausea. Get over it and focus on the long-term goal of cell membrane stability. If you can't handle the titration, you'll never handle the disease. Just follow the science and stop making excuses for your lack of progress. It's really not that hard if you actually listen to someone who knows what they're talking about. Seriously, just do the work.
typical weak mindset thinking a pill is the only way out
you need to strengthen your spirit and your blood the american way and stop relying on these foreign-sounding biologics
It is truly commendable that such detailed information is being shared to assist those who have struggled for years to find a diagnosis. The emphasis on the distinction between antihistamines and stabilizers provides a necessary perspective for patient education.
I really hope everyone reading this finds the relief they deserve. It's a long road but you aren't alone in this struggle.
Stay strong everyone. You can do this.
too much reading for a basic summary