Tag: FDA
The Purple Book: Understanding Biosimilars and Interchangeability from the FDA
- Benjamin Aghaki-Allen
- Health
- 15 comment
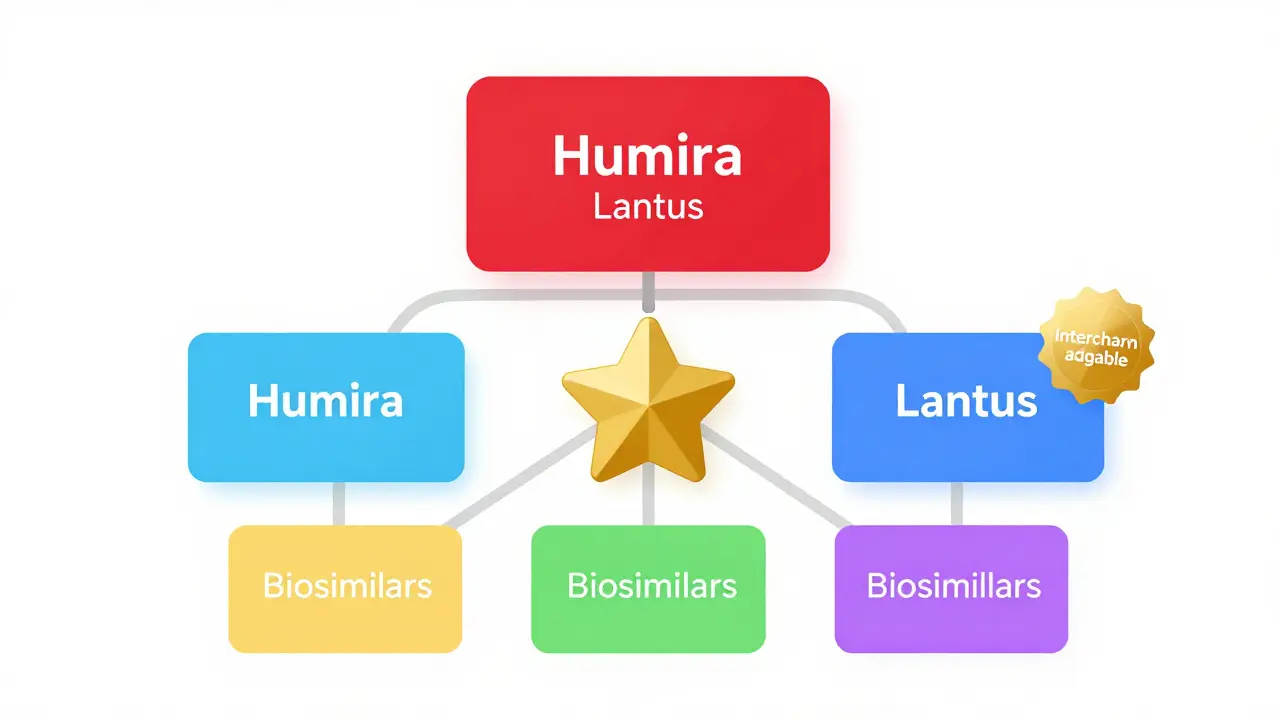
The FDA's Purple Book is the official guide to biosimilars and interchangeable biological products. Learn how it helps patients and pharmacists understand substitution rules, regulatory status, and key differences between biosimilars and interchangeable versions.
VIEW MORECategories
- Health (173)
- Health & Wellness (7)
- Pharmaceutical Research (5)
- Nutrition & Weight Loss (4)
- Climate & Environment (1)
Popular posts
-
How to Transition from Hospital to Home Without Medication Errors
Benjamin Aghaki-Allen -
How Cefadroxil Works to Fight Bacterial Infections - Mechanism, Uses & Safety
Benjamin Aghaki-Allen -
Antibiotic & Birth Control Pill Interactions: Proven Facts
Benjamin Aghaki-Allen -
Blond Psyllium Daily: Benefits, Dosage, and How to Use for Digestive Health
Benjamin Aghaki-Allen -
How and Where to Buy Indocin Online Safely (2025 Guide)
Benjamin Aghaki-Allen
Popular tags
- medication safety
- medication adherence
- side effects
- generic drugs
- online pharmacy
- medication errors
- drug side effects
- bioequivalence
- drug interactions
- dietary supplement
- gut health
- blood pressure medication
- alternatives
- senior medications
- Hatch-Waxman Act
- generic medications
- adverse drug reactions
- medication reconciliation
- generic substitution
- narrow therapeutic index